|
Back to Blog
Cathode vs anode5/7/2023 
Anode and cathode are defined by this current flow. ‘Current-out’ means current is flowing out of the system. For a device, when we say ‘current-in,’ that means the current is flowing into the system. When the electrons are flowing to one direction, we say current is flowing to the opposite direction to the electrons. In other words, current is carried by moving electrons. When a current is flowing, negatively charged electrons are flowing. Electrochemical cells, cathode ray tubes, and X-ray tubes are some examples where we come across anodes and cathodes. 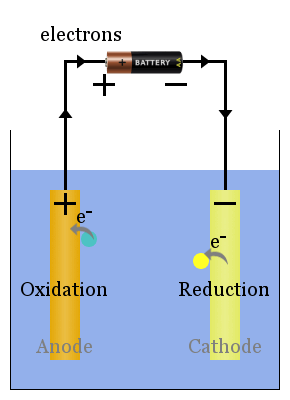
MICHAEL CARANO is vice president for OM Group, Inc.Anode and cathode are necessary for electrical set ups where current flow is involved. By mitigating the effects of these resistances, plating Plating distribution across the surface of a printed wiring board panel,Īnd from the surface of the panel through the holes, it will vary due to Will ensure uniform plating distribution. There are no guarantees that uniform anode polarization SolutionĪgitation at the anodes will minimize both concentration and gas Variations from the top of the plated panel to the bottom. Polarization will cause the anodes to sludge causing thickness Planar equi-potential surface is radiated from the anode. Polarization to insure that the anodes are uniformly polarized and a One variable may have a number of effects. Secondary current distribution effects are complicated. TheĬoncentration polarization can be altered by solution agitation. Ions are depleted from solution close to the surface of the cathode. Finally, concentration polarization results as the metal High a concentration of the metal salt in solution in combination with Temperature, imbalance of key addition agents or a combination of too Plugged with sludge that has fallen from the anodes and requiresĬleaning and operating parameters of the plating process that can lead A number of factorsĬontribute to this issue: poor quality anodes, anode bags that are One sees a thick sludge-like film form on the anode. 
Chemical polarization results from a thick filmįorming on the anodes, which results in poor anode corrosion. Movement), and more importantly solution movement uniformity, gas By increasing solution agitation (solution Gas polarization results directly from oxygen and hydrogen being evolvedĭuring the plating process. Gas polarization, concentration polarization and chemical polarization. The three types of polarization encountered in electroplating are Solution operating temperature, solution agitation and cathode current Polarization and conductivity are both dependent on Increase the conductivity of the plating electrolyte and the In order to improve throwing power and plating uniformity, one can Reduce the metal ions to metal at the cathode. The voltage required to corrode the anode and the resistance required to Is determined by Ohm's Law-which is the solution resistanceīetween the anodes and cathodes. Potential exists between the anode and cathode. When a plating cell contains copper anodes and aĬopper cathode (circuit board) in a plating electrolyte, an equilibrium Required above the equilibrium potential to drive the deposition of the Polarization refers to the additional potential Rack design and cathode spacing on the rack will also influence theįortunately, secondary current comes into play through a factor That anode to cathode surface area ratios should not exceed 2:1 andĪnode length should be three to six inches shorter than the cathode. To minimize the primary current distribution, it is recommended Pattern plating of PCBs represents a similar situation. Plated to a much greater thickness than other areas, as depicted inįIGURE 2. Increased current flow around the edges of the panels (cathode) will be If the anodes are much larger in area than the cathode, FIGURE 1 illustrates theĪnode surface area versus cathode surface area is another importantĬonsideration. Increasing the anode toĬathode distance minimizes plating variations. Is reduced and plating uniformity suffers. OFTEN, IN AN EFFORT to increase throughput, anode/ cathode spacing APA style: Electroplating of copper, Part 4: anode-cathode placement and secondary current distribution.Electroplating of copper, Part 4: anode-cathode placement and secondary current distribution." Retrieved from MLA style: "Electroplating of copper, Part 4: anode-cathode placement and secondary current distribution." The Free Library.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
